Breaking Window Bars with a Bicycle
This is another classic MacGyver hack. There is a window with bars on the outside and Mac needs to get IN. Simple, just pull off the bars. Obviously you can’t do this with your hands, you need to build something.
I guess you would call this a hand-crank winch. That probably best describes what he builds. Actually, it’s a hand crank winch WITH a compound pulley. Here are a couple of pictures.


The main idea here is the same for ALL simple machines. It’s really about force, distance and work. Let’s start with a super basic definition of work (physics work).
In this expression (which isn’t technically correct—but that’s OK for now), W is the work, F is the force applied and d is the distance over which the force moves.
Now imagine I have a simple machine. I can put work into it and get work out of it. Assuming it is 100 percent efficient, the work in can’t be less than the work out (or you would get FREE ENERGY).
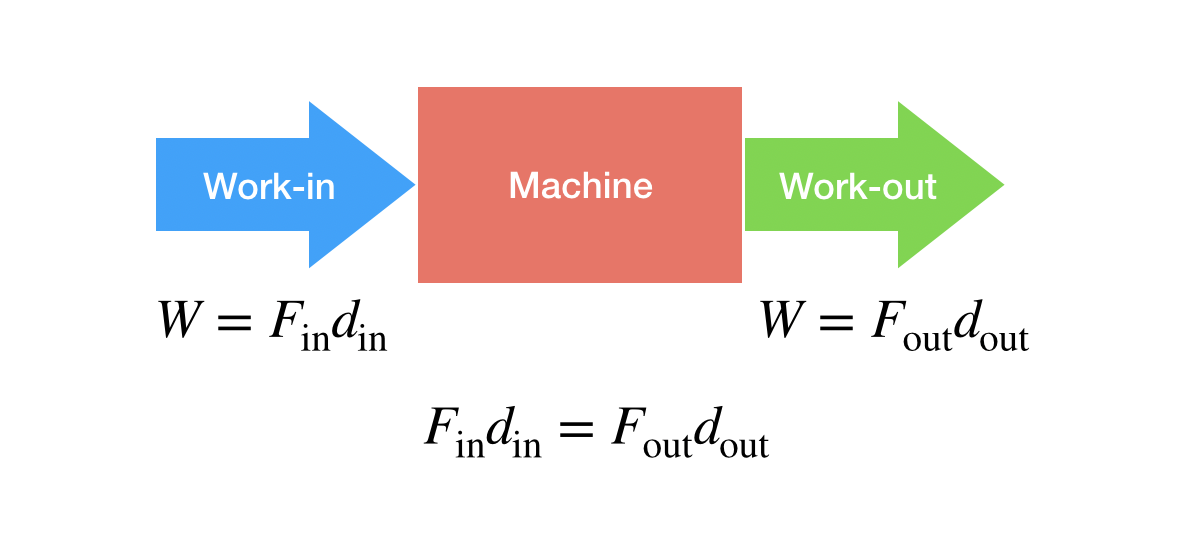
So, if you want to pull (or push) with a smaller force then you need to pull over a larger distance. For the output force, you need to have it move over a shorter distance to get a larger force.
With the winch, MacGyver turns a hand crank (part of the bike). If the garden hose (used for a rope) is wrapping around something with a smaller diameter, then for each rotation of the hand the rope will move a small distance. This is the key to a winch. Remember—smaller distance means larger force.
With the compound pulley, the same thing happens. By using multiple pulleys—you can make the pull force move over a larger distance thus increasing the output force.
Here is my initial diagram for how this might work.

Spark Gap Generator
You don’t get to see much detail here—so let me just explain the idea behind a spark gap generator.
First, I guess I should say what it is used for. Originally, a spark gap was a radio transmitter. It turns out that although it’s simple to build, you can only use one at a time because they don’t really use channels. In the case of MacGyver, he is using a spark gap to jam a phone signal (to prevent data transfer).
All of the wireless data works by broadcasting and receiving electromagnetic waves. Radio, microwave, visible light, x-rays…these are all electromagnetic waves (but with different wavelengths). Still, they are electromagnetic waves.
So, what about this spark gap? The idea is to create a repeating spark across some small gap. This spark is a very violent (electromagnetically speaking) event. It has accelerating electric charges which create EM waves. These EM waves are high enough intensity that they can make it such that other (more well behaved) devices can’t send or receive a signal.
But how do you make one of these spark gap generators? Really, you just need a battery and some wires. If you use the wires and battery you can create an electromagnet. That doesn’t make a spark, but if you can turn it on and off really quickly, then it will indeed make a spark. I built one using a moving metal switch. When the electromagnet is on, it pulls the metal and turns off the switch. Once the switch is off, the metal is no longer attracted to the electromagnet and it moves back in place to turn the electric current on again. This just repeats to make the spark.
Here is a video.
Here are some more details on this.
Oh, here is another way to make one of these spark gap generators.
Gum Wrapper Switch
The key to this episode (it’s in the title) is gum. MacGyver activates the spark gap by taking out a piece of gum. How would this work?
The purpose of a switch is to do something such that two wires are connected. In this case, the two ends of the circuit could be the foil wrappers for two pieces of gum. If you put an insulator (gum) in between them, then the circuit will be closed. Pull the gum out and then two foil pieces will touch and complete the circuit.
Here is a diagram I created for this.
